AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Pauli exclusion principle d1/23/2024 Since each electron must maintain its unique identity, we intuitively sense that the four quantum numbers for any given electron must not match up exactly with the four. The Pauli exclusion principle says that no two electrons can have the same set of quantum numbers that is, no two electrons can be in the same state. 
N2 - "Pauli's Exclusion Principle proposes a philosophical framework for understanding the principle's origin in the atomic spectroscopy of the early 1920s, its subsequent embedding in quantum mechanics and later experimental validation with the development of quantum chromodynamics."-BOOK JACKETĪB - "Pauli's Exclusion Principle proposes a philosophical framework for understanding the principle's origin in the atomic spectroscopy of the early 1920s, its subsequent embedding in quantum mechanics and later experimental validation with the development of quantum chromodynamics. When we look at the orbital possibilities for a given atom, we see that there are different arrangements of electrons for each different type of atom. Let us consider a system of two electrons, with wavefunctions, A (x 1) and B (x 2). The Exclusion principle is a consequence of two simple facts: electrons are fermions and they are indistinguishable. This means that two electrons in a single orbital must have opposite spins. An explanation of any Pauli repulsion model must start with a basic understanding of the Pauli exclusion principle. N1 - Includes bibliographical references and index First, the Pauli exclusion principle lets us know that no two electrons can have the exact same quantum numbers. This is the main difference between Pauli exclusion. 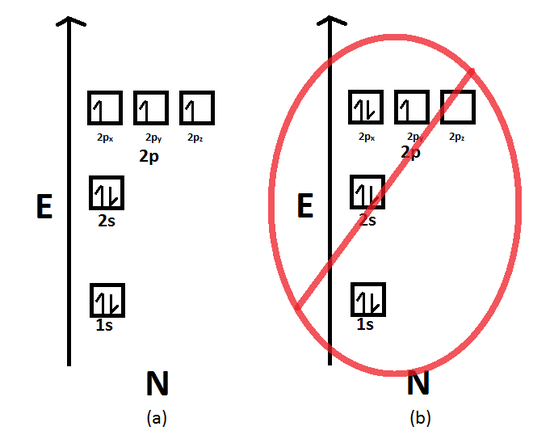
The usual statement of the Pauli Exclusion Principle is that no two electrons in an atom can have the same set of four quantum numbers n, l. In other words, (1) no more than two electrons can occupy the same orbital and (2) two electrons in the same orbital must have opposite spins (Figure 46(i) and (ii)). Pauli Exclusion Principle focuses on the unique identification and pairing of individual electrons within orbitals, while Hund’s Rule addresses the arrangement of electrons within degenerate orbitals, emphasizing the tendency of electrons to occupy orbitals singly before pairing up. One explanation as to why the differences between the term symbols that arise from a p2 p 2 configuration relative to a pp configuration is the Pauli Exclusion principle. For the Pauli exclusion principle, just remember. Pauli's Exclusion Principle states that no two electrons in the same atom can have identical values for all four of their quantum numbers. Hund's rule explains why each orbital gets an electron before any orbital has two electrons, while Pauli explains why an orbital can't have more than two electrons (since there are only two spin states). To investigate the consequences of ignoring the PEP, we discuss several algebraic models in nuclear structure physics, in particular cluster models. Consistent with Pauli’s exclusion principle. The Pauli Exclusion Principle (PEP) is one of the most basic concepts in physics, but also the most difficult to implement in many-fermion systems, which are common in nuclear physics. Sample sets of quantum numbers for the electrons in an atom are given in Table 8.5.1 8.5. Q: What is Pauli exclusion principleAns: Pauli exclusion principle is def. This principle is related to two properties of electrons: All electrons are identical (when you’ve seen one electron, you’ve seen them all) and they have half-integral spin ( s 1/2 s 1 / 2 ). 14 Importance Of Pauli Exclusion Principle If this principle were not valid, an atom could radiate energy until every electron in the atom is in the lowest possible energy state and therefore the chemical behavior of the elements would be grossly modified. \) spin) to the external magnetic and fill first.T2 - the origin and validation of a scientific principle The Pauli exclusion principle states that an orbital cannot have electrons of the same spin. This lecture is about Pauli exclusion principle and spin of electrons in orbitals.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
